Oxidation is the gain of oxygen by a substance. For example, magnesium is oxidised when it reacts with oxygen to form magnesium oxide:
magnesium + oxygen → magnesium oxide
2Mg + O2→ 2MgO
Reduction is the loss of oxygen from a substance. For example, copper oxide can be reduced to form copper if it is reacted with hydrogen:
copper oxide + hydrogen → copper + water
CuO + H2→ Cu + H2O
Many ores contain metal oxides, therefore many metals can be extracted from their ores by reduction reactions. The method used to extract a given metal depends on how reactive it is:
iron + water + oxygen → hydrated iron(III) oxide
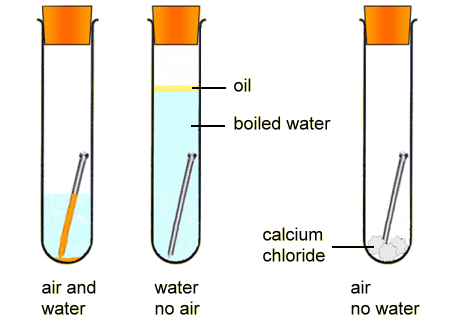
In the experiment below, the nail does not rust when air - containing oxygen - or water is not present:
magnesium + oxygen → magnesium oxide
2Mg + O2→ 2MgO
Reduction is the loss of oxygen from a substance. For example, copper oxide can be reduced to form copper if it is reacted with hydrogen:
copper oxide + hydrogen → copper + water
CuO + H2→ Cu + H2O
Many ores contain metal oxides, therefore many metals can be extracted from their ores by reduction reactions. The method used to extract a given metal depends on how reactive it is:
- very reactive metals – electrolysis
- less reactive metals - reduction
iron + water + oxygen → hydrated iron(III) oxide
In the experiment below, the nail does not rust when air - containing oxygen - or water is not present:
Salt dissolved in water does not cause rusting, but it does speed it up, as does acid rain.
Aluminium does not rust (corrode) because its surface is protected by a natural layer of aluminium oxide which prevents the metal below from coming into contact with air and oxygen. Unlike rust, which can flake off the surface of iron and steel objects, the layer of aluminium oxide does not flake off.
More reactive elements are more likely to oxidise.
Aluminium does not rust (corrode) because its surface is protected by a natural layer of aluminium oxide which prevents the metal below from coming into contact with air and oxygen. Unlike rust, which can flake off the surface of iron and steel objects, the layer of aluminium oxide does not flake off.
More reactive elements are more likely to oxidise.